Section 1.5 Experiments
¶In the last section we investigated observational studies and sampling strategies. While these are effective tools for answering certain research questions, often times researchers want to measure the effect of a treatment. In this case, they must carry out an experiment. Just as randomization is essential in sampling in order to avoid selection bias, randomization is essential in the context of experiments to determine which subjects will receive which treatments. If the researcher chooses which patients are in the treatment and control groups, she may unintentionally place healthier or sicker patients in one group or the other, biasing the experiment either for or against the treatment.
OpenIntro: Experiments video
Subsection 1.5.1 Reducing bias in human experiments
¶Randomized experiments are essential for investigating cause and effect relationships, but they do not ensure an unbiased perspective in all cases. Human studies are perfect examples where bias can unintentionally arise. Here we reconsider a study where a new drug was used to treat heart attack patients. 1 Anturane Reinfarction Trial Research Group. 1980. Sulfinpyrazone in the prevention of sudden death after myocardial infarction. New England Journal of Medicine 302(5):250-256. In particular, researchers wanted to know if the drug reduced deaths in patients.
These researchers designed a randomized experiment because they wanted to draw causal conclusions about the drug's effect. Study volunteers 2 Human subjects are often called patients, volunteers, or study participants. were randomly placed into two study groups. One group, the treatment group, received the drug. The other group, called the control group, did not receive any drug treatment. In an experiment, the explanatory variable is also called a factor. Here the factor is receiving the drug treatment. It has two levels: yes and no, thus it is categorical. The response variable is whether or not patients died within the time frame of the study. It is also categorical.
Put yourself in the place of a person in the study. If you are in the treatment group, you are given a fancy new drug that you anticipate will help you. On the other hand, a person in the other group doesn't receive the drug and sits idly, hoping her participation doesn't increase her risk of death. These perspectives suggest there are actually two effects: the one of interest is the effectiveness of the drug, and the second is an emotional effect that is difficult to quantify.
Researchers aren't usually interested in the emotional effect, which might bias the study. To circumvent this problem, researchers do not want patients to know which group they are in. When researchers keep the patients uninformed about their treatment, the study is said to be blind or single-blind. But there is one problem: if a patient doesn't receive a treatment, she will know she is in the control group. The solution to this problem is to give fake treatments to patients in the control group. A fake treatment is called a placebo, and an effective placebo is the key to making a study truly blind. A classic example of a placebo is a sugar pill that is made to look like the actual treatment pill. Often times, a placebo results in a slight but real improvement in patients. This effect has been dubbed the placebo effect.
The patients are not the only ones who should be blinded: doctors and researchers can accidentally bias a study. When a doctor knows a patient has been given the real treatment, she might inadvertently give that patient more attention or care than a patient that she knows is on the placebo. To guard against this bias, which again has been found to have a measurable effect in some instances, most modern studies employ a double-blind setup where researchers who interact with subjects and are responsible for measuring the response variable are, just like the subjects, unaware of who is or is not receiving the treatment. 3 There are always some researchers involved in the study who do know which patients are receiving which treatment. However, they do not interact with the study's patients and do not tell the blinded health care professionals who is receiving which treatment.
Look back to the study in Section 1.1 where researchers were testing whether stents were effective at reducing strokes in at-risk patients. Is this an experiment? Was the study blinded? Was it double-blinded? 4 The researchers assigned the patients into their treatment groups, so this study was an experiment. However, the patients could distinguish what treatment they received, so this study was not blind. The study could not be double-blind since it was not blind.
Subsection 1.5.2 Principles of experimental design
¶Well-conducted experiments are built on three main principles.
-
Direct Control. Researchers assign treatments to cases, and they do their best to control any other differences in the groups. They want the groups to be as identical as possible except for the treatment, so that at the end of the experiment any difference in response between the groups can be attributed to the treatment and not to some other confounding or lurking variable. For example, when patients take a drug in pill form, some patients take the pill with only a sip of water while others may have it with an entire glass of water. To control for the effect of water consumption, a doctor may ask all patients to drink a 12 ounce glass of water with the pill.
Direct control refers to variables that the researcher can control, or make the same. A researcher can directly control the appearance of the treatment, the time of day it is taken, etc. She cannot directly control variables such as gender or age. To control for these other types of variables, she might consider blocking, which is described in Subsection 1.5.3.
Randomization. Researchers randomize patients into treatment groups to account for variables that cannot be controlled. For example, some patients may be more susceptible to a disease than others due to their dietary habits. Randomizing patients into the treatment or control group helps even out the effects of such differences, and it also prevents accidental bias from entering the study.
Replication. The more cases researchers observe, the more accurately they can estimate the effect of the explanatory variable on the response. In an experiment with six subjects, even if there is randomization, it is quite possible for the three healthiest people to be in the same treatment group. In a randomized experiment with 100 people, it is virtually impossible for the healthiest 50 people to end up in the same treatment group. In a single study, we replicate by imposing the treatment on a sufficiently large number of subjects or experimental units. A group of scientists may also replicate an entire study to verify an earlier finding. However, each study should ensure a sufficiently large number of subjects because, in many cases, there is no opportunity or funding to carry out the entire experiment again.
It is important to incorporate these design principles into any experiment. If they are lacking, the inference methods presented in the following chapters will not be applicable and their results may not be trustworthy. In the next section we will consider three types of experimental design.
Subsection 1.5.3 Completely randomized, blocked, and matched pairs design
¶
A completely randomized experiment is one in which the subjects or experimental units are randomly assigned to each group in the experiment. Suppose we have three treatments, one of which may be a placebo, and 300 subjects. To carry out a completely randomized design, we could randomly assign each subject a unique number from 1 to 300, then subjects with numbers 1-100 would get treatment 1, subjects 101-200 would get treatment 2, and subjects 201- 300 would get treatment 3. Note that this method of randomly allocating subjects to treatments in not equivalent to taking a simple random sample. Here we are not sampling a subset of a population; we are randomly splitting subjects into groups.
While it might be ideal for the subjects to be a random sample of the population of interest, that is rarely the case. Subjects must volunteer to be part of an experiment. However, because randomization is incorporated in the splitting of the groups, we can still use statistical techniques to check for a causal connection, though the precise population for which the conclusion applies may be unclear. For example, if an experiment to determine the most effective means to encourage individuals to vote is carried out only on college students, we may not be able to generalize the conclusions of the experiment to all adults in the population.
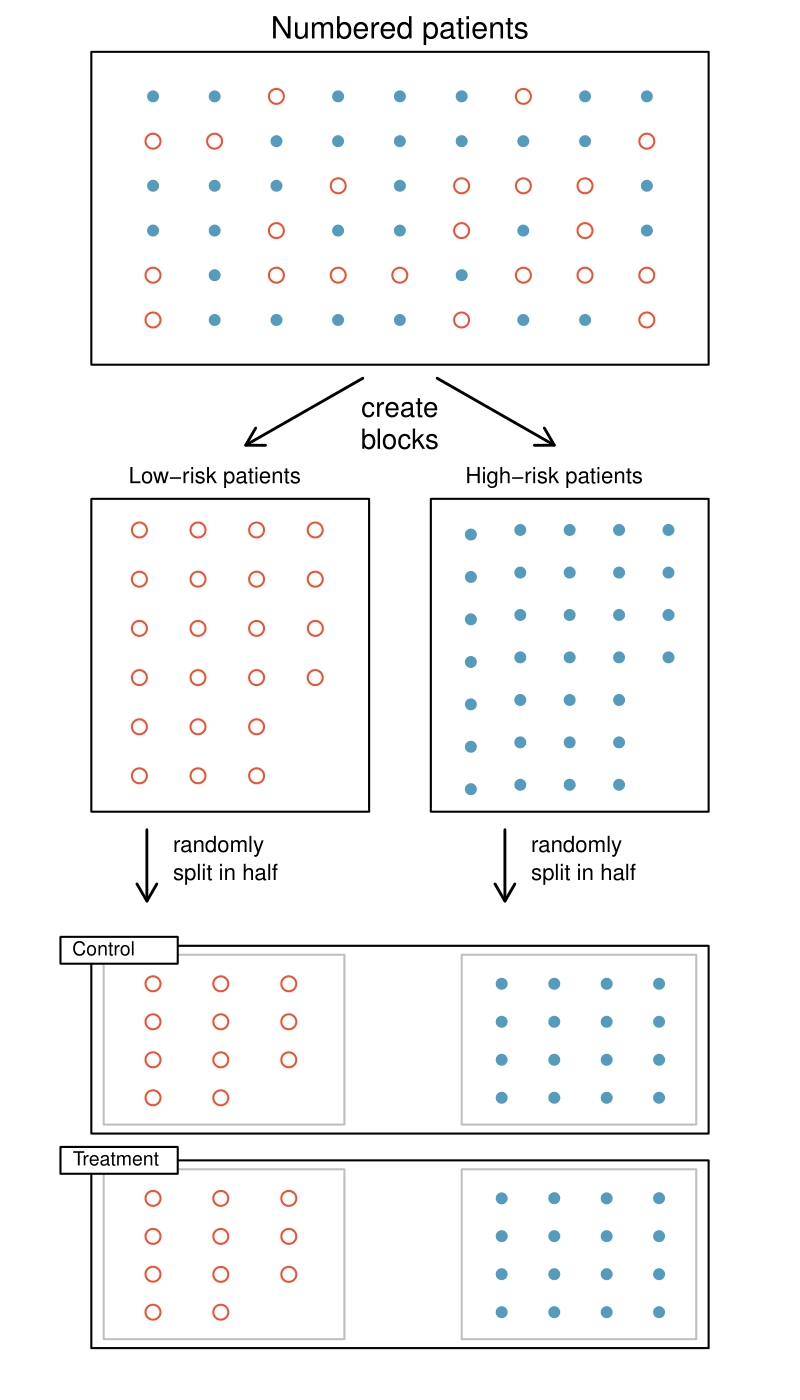
Researchers sometimes know or suspect that another variable, other than the treatment, influences the response. Under these circumstances, they may carry out a blocked experiment. In this design, they first group individuals into blocks based on the identified variable and then randomize subjects within each block to the treatment groups. This strategy is referred to as blocking. For instance, if we are looking at the effect of a drug on heart attacks, we might first split patients in the study into low-risk and high-risk blocks. Then we can randomly assign half the patients from each block to the control group and the other half to the treatment group, as shown in Figure 1.5.3. At the end of the experiment, we would incorporate this blocking into the analysis. By blocking by risk of patient, we control for this possible confounding factor. Additionally, by randomizing subjects to treatments within each block, we attempt to even out the effect of variables that we cannot block or directly control.
Example 1.5.4
An experiment will be conducted to compare the effectiveness of two methods for quitting smoking. Identify a variable that the researcher might wish to use for blocking and describe how she would carry out a blocked experiment.
The researcher should choose the variable that is most likely to influence the response variable - whether or not a smoker will quit. A reasonable variable, therefore, would be the number of years that the smoker has been smoking. The subjects could be separated into three blocks based on number of years of smoking and each block randomly divided into the two treatment groups.
Even in a blocked experiment with randomization, other variables that affect the response can be distributed unevenly among the treatment groups, thus biasing the experiment in one direction. A third type of design, known as matched pairs addresses this problem. In a matched pairs experiment, pairs of people are matched on as many variables as possible, so that the comparison happens between very similar cases. This is actually a special type of blocked experiment, where the blocks are of size two.
An alternate form of matched pairs involves each subject receiving both treatments. Randomization can be incorporated by randomly selecting half the subjects to receive treatment 1 first, followed by treatment 2, while the other half receives treatment 2 first, followed by treatment.
Guided Practice 1.5.5
How and why should randomization be incorporated into a matched pairs design? 5 Assume that all subjects received treatment 1 first, followed by treatment 2. If the variable being measured happens to increase naturally over the course of time, it would appear as though treatment 2 had a greater effect than it really did.
Guided Practice 1.5.6
Matched pairs sometimes involves each subject receiving both treatments at the same time. For example, if a hand lotion was being tested, half of the subjects could be randomly assigned to put Lotion A on the left hand and Lotion B on the right hand, while the other half of the subjects would put Lotion B on the left hand and Lotion A on the right hand. Why would this be a better design than a completely randomized experiment in which half of the subjects put Lotion A on both hands and the other half put Lotion B on both hands? 6 The dryness of people's skins varies from person to person, but probably less so from one person's right hand to left hand. With the matched pairs design, we are able control for this variability by comparing each person's right hand to her left hand, rather than comparing some people's hands to other people's hands (as you would in a completely randomized experiment).
Because it is essential to identify the type of data collection method used when choosing an appropriate inference procedure, we will revisit sampling techniques and experiment design in the subsequent chapters on inference.
Subsection 1.5.4 Testing more than one variable at a time
Some experiments study more than one factor (explanatory variable) at a time, and each of these factors may have two or more levels (possible values). For example, suppose a researcher plans to investigate how the type and volume of music affect a person's performance on a particular video game. Because these two factors, type and volume, could interact in interesting ways, we do not want to test one factor at a time. Instead, we want to do an experiment in which we test all the combinations of the factors. Let's say that volume has two levels (soft and loud) and that type has three levels (dance, classical, and punk). Then, we would want to carry out the experiment at each of the six (2 x 3 = 6) combinations: soft dance, soft classical, soft punk, loud dance, loud classical, loud punk. Each of the these combinations is a treatment. Therefore, this experiment will have 2 factors and 6 treatments. In order to replicate each treatment 10 times, one would need to play the game 60 times.
Guided Practice 1.5.7
A researcher wants to compare the effectiveness of four different drugs. She also wants to test each of the drugs at two doses: low and high. Describe the factors, levels, and treatments of this experiment. 7 There are two factors: type of drug, which has four levels, and dose, which has 2 levels. There will be 4 x 2 = 8 treatments: drug 1 at low dose, drug 1 at high dose, drug 2 at low dose, and so on.
As the number of factors and levels increases, the number of treatments become large and the analysis of the resulting data becomes more complex, requiring the use of advanced statistical methods. We will investigate only one factor at a time in this book.
